Expanded Medicare Telehealth Coverage for Opioid Use Disorder Treatment Services Furnished by Opioid Treatment Programs
Healthcare Law Today
NOVEMBER 9, 2022
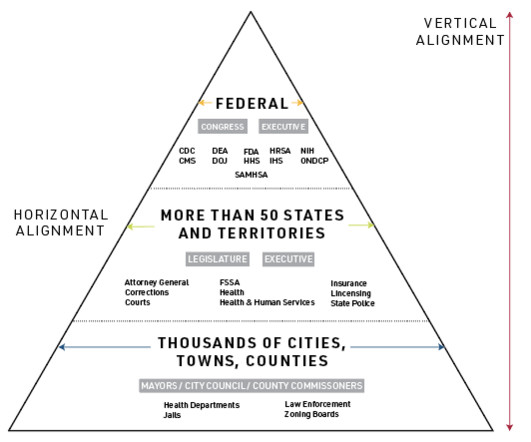
Starting January 1, 2023, Medicare will cover telehealth-based treatment services delivered by federally-accredited opioid treatment programs (OTPs), commonly referred to as “methadone clinics.” CMS also extended coverage for SUD treatment services provided via telehealth.















Let's personalize your content