FDA Selects Ontada to Investigate Rare Cancers Treated in US Community Oncology Setting
HIT Consultant
OCTOBER 11, 2023
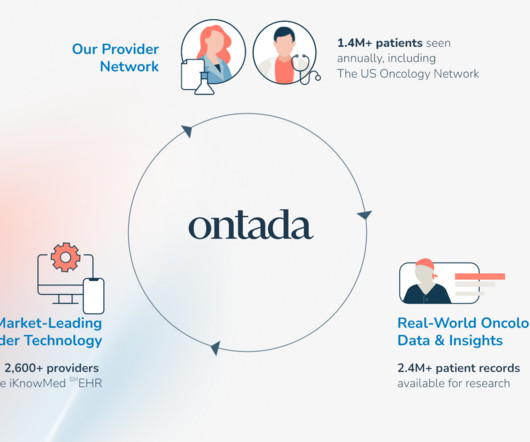
Food and Drug Administration (FDA) to advance the use of real-world data (RWD) in the U.S. Study Goals & Objectives The goal of the study is to better understand how patients with rare cancers present and are treated to inform drug development for life sciences companies and the FDA. community oncology setting.











































Let's personalize your content