Litigation Challenges Prioritization of Race or Ethnicity in Allocating COVID-19 Therapies
Bill of Health
MARCH 28, 2022
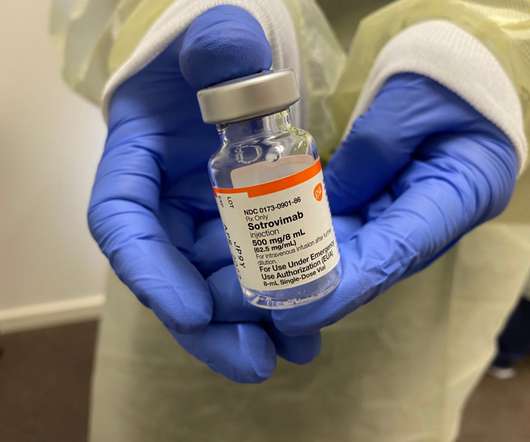
By James Lytle. Recent guidance from the U.S. Food & Drug Administration (FDA) encouraged several states to adopt policies that prioritized race or ethnicity in the allocation of monoclonal antibody treatments and oral antivirals for the treatment of SARS-CoV-2. The guidance proved to be highly controversial, prompting two states, Utah and Minnesota, to withdraw their guidance , and leading a third state, New York, to become the subject of two federal lawsuits that challenge the guidance’s l







































Let's personalize your content